Investment Case
The world's first fully implantable, closed-loop BCI in clinical use.
Building the standard of care in neurotherapy.
Why now?
First-in-human milestone achieved, clinical momentum accelerating, market leadership position still available.
Why Brain Interchange?
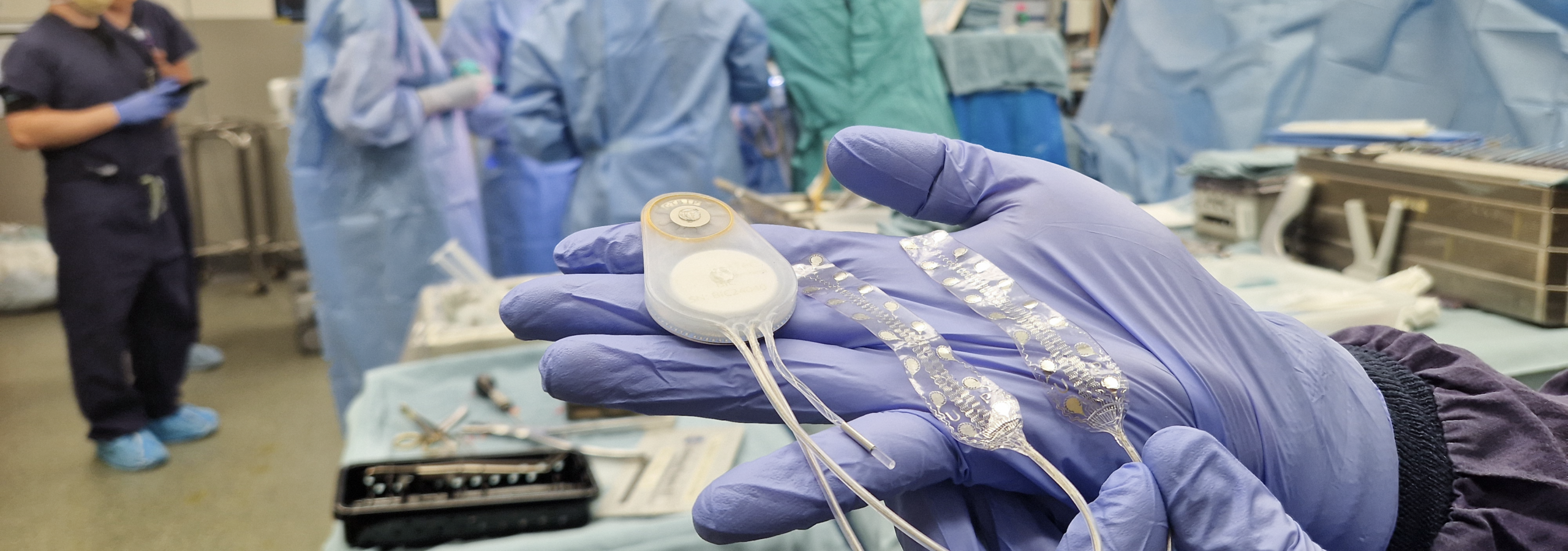
The only fully implantable, wireless streaming, closed-loop BCI in clinical use. Entirely developed and manufactured in Germany. 10+ years of human neural data across 4 indications and 50+ electrode implantations.
Why this deal?
We operate in a fast‑growing global neurotechnology market and are strategically positioned to shape its future. We are seeking partners and investors to join us in building this forward‑looking, innovation‑driven equity story.
Market Opportunity
Leading analysts have identified neurotechnology as the defining MedTech opportunity of the decade.
“The next big opportunity in MedTech – and it’s rare.”
Morgan Stanley, October 2024
$48B
Beachhead Stroke Rehabilitation (US)
$240B
Expansion: Depression (US)
$400B
Total BCI Market by 2045*
Clinical Milestones
July 2025 – First-in-human Implantation, UW Medicine, Seattle, USA
February 2026 – Successful implantation of the Brain Interchange in Patient 2 of the study.
Q2 2026 – planned implantation of Patient 3 of the Study.
The Brain Interchange System is an investigational device and is not approved for commercial use. CAUTION: Investigational device. Limited by Federal (or United States) law to investigational use.
Clinical Derisking
Stronger Motor Recovery in 6 Weeks Than in the Entire Previous Year
Observed in first study participant. Investigational device.
Therapeutic Effect Persists After Stimulation Stops
Observed in first study participant. Investigational device.
Stable Wireless Operation for 6+ Months
Observed in first study participant. Investigational device.