Clinical Pathway
Brain Interchange™ is designed to restore movement after neurological injury by creating a direct communication channel between the brain and external rehabilitation technologies. In stroke survivors, motor intention signals can be decoded and transformed into functional actions, such as muscle activation or control of assistive devices, supporting intensive, task‑oriented rehabilitation.
The goal: to enable more effective, personalized recovery for individuals living with motor impairments after stroke.
Stronger Motor Recovery in 6 Weeks Than in the Entire Previous Year
Therapeutic Effect Persists After Stimulation Stops
Stable Wireless Operation for 6+ Months
The U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for CorTec’s Brain Interchange™ system. The designation covers the use of direct cortical electrical stimulation to support motor recovery in people living with chronic stroke-related impairments.

CTO Martin Schuettler explains the technology and clinical progress behind the first two human implantations of the Brain Interchange system, performed at Harborview Medical Center in Seattle as part of an NIH-funded, FDA-approved IDE Study. He reflects on what the early results mean for stroke rehabilitation and the road ahead.
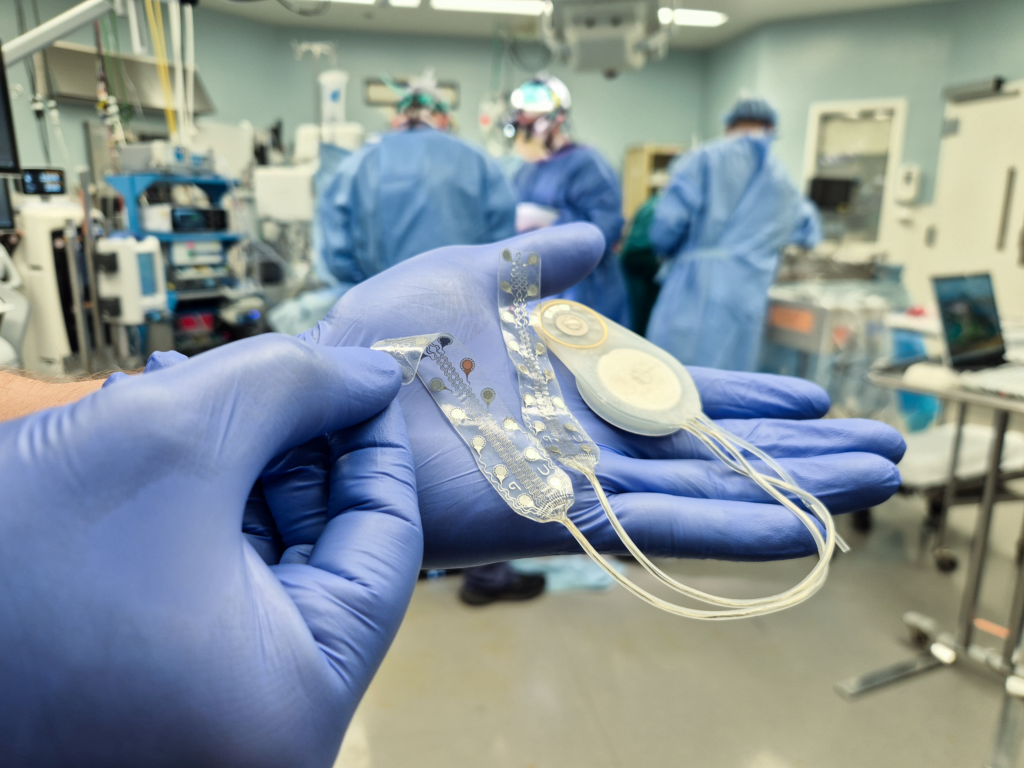
In February 2026, a second patient at Harborview Medical Center in Seattle successfully received the Brain Interchange™ implant as part of an NIH-funded, FDA-approved trial led by researchers from the University of Washington and UCLA. Building on encouraging results from the first participant, the study continues to advance CorTec’s clinical program for stroke recovery.
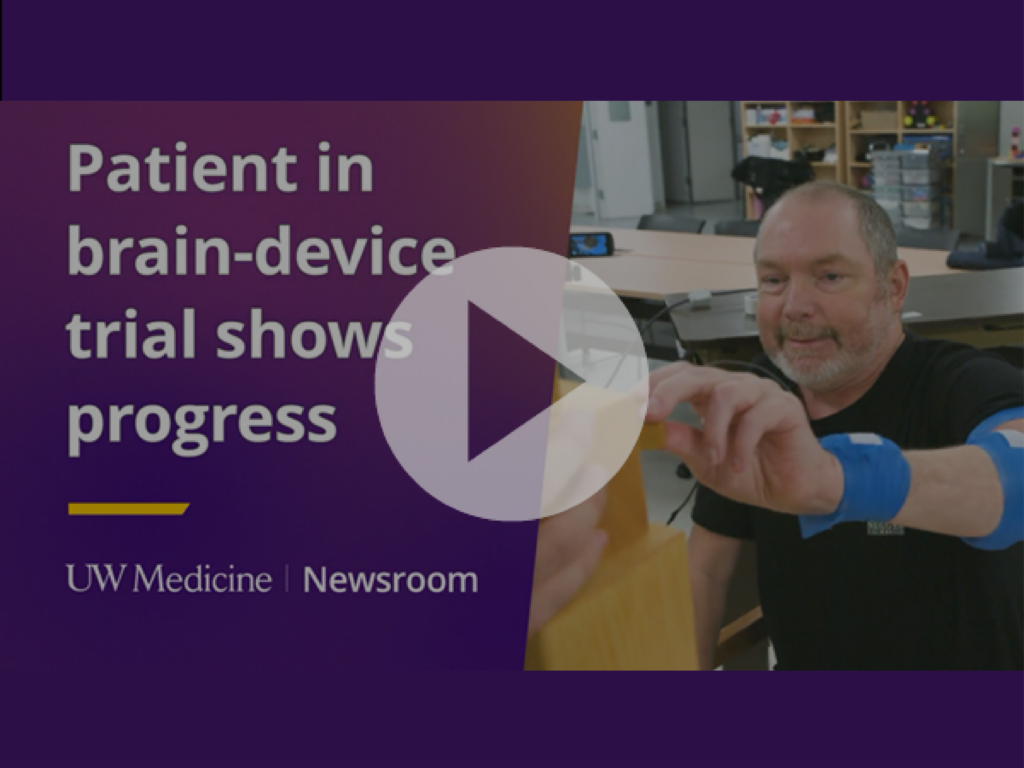
In December 2025, the University of Washington shared the story of Matt Kidd, a stroke patient who regained motor function as part of a clinical trial using a brain implant developed in collaboration with CorTec. After years of therapy with little progress, targeted electrical stimulation helped his nerve cells reconnect and within weeks he was able to perform everyday movements again.
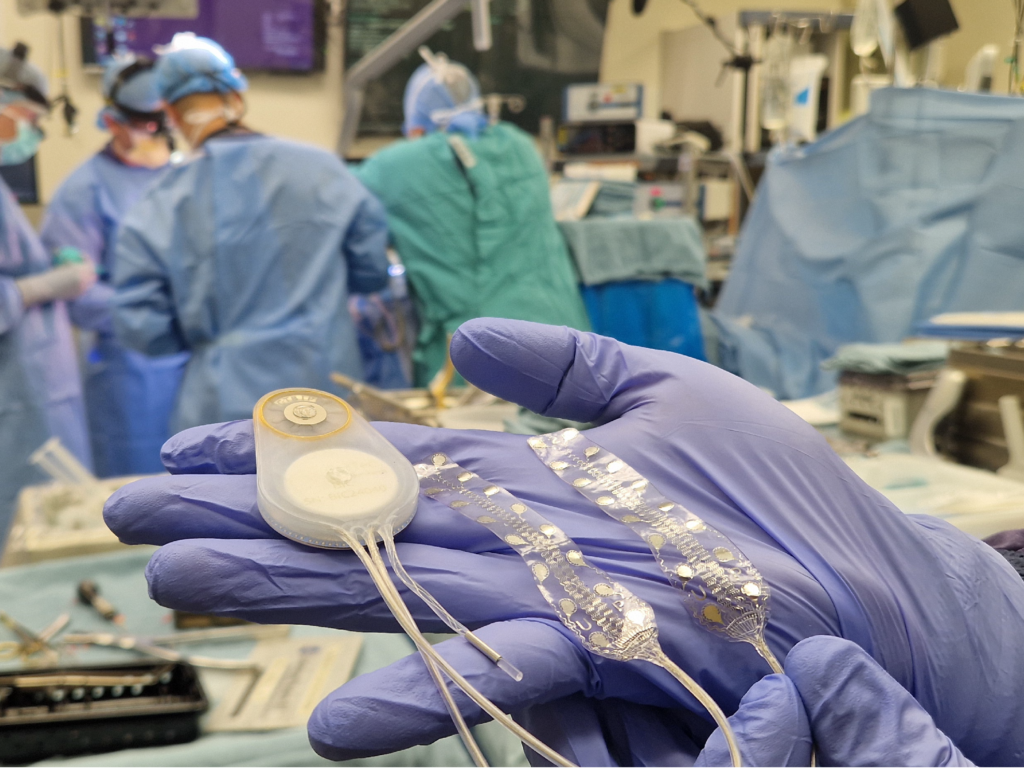
In July 2025, CorTec achieved a historic milestone with the first-ever human implantation of a fully implantable, German-developed Brain-Computer Interface system, performed at Harborview Medical Center in Seattle. As part of an FDA-approved trial led by researchers from the University of Washington and UCLA, the study targets motor recovery in stroke patients and marks a major step forward in neurorehabilitation.

In October 2024, CorTec received both FDA Investigational Device Exemption and IRB approval from the University of Washington, clearing the path for the first-ever human implantation of a fully implantable, German-developed Brain-Computer Interface system. The study, conducted in collaboration with researchers from the University of Washington and UCLA, targets motor recovery in stroke patients with arm paralysis.
Mayo Clinic collaboration
Led by Prof. Nuri Ince and Prof. Gregory Worrell.
University Clinic of Freiburg
First-in-Human study in planning
We work directly with clinicians and researchers to co-design each BCI system. Your specific indication, patient population, and therapeutic protocol determine the exact configuration, from electrode placement to stimulation algorithms.